Production method
Carbide is produced in electric furnaces by fusing (calcining) a mixture of coke and calcium oxide (quicklime) at temperatures ranging from 1900°C to 2300°C. The sharp and unpleasant garlic smell of carbide and the produced acetylene is caused by the content of impurities of calcium phosphides and sulfates in the carbide mixture.
The process is carried out in several stages:
- Limestone is fired.
- A powder mixture is created from processed lime and coke - a mixture.
- The resulting mixture is calcined in an electric arc furnace until it melts.
- The resulting carbide bars are crushed to obtain the desired fraction.
The average density of the carbide substance is 2.2 g/cm3. Depending on the impurity content, the color of carbide can be dark brown or dark gray.
The final product consists of 75-78% CaC2, the rest being lime and impurities. Carbide granules come in different sizes: 2x8; 8x15; 15×25; 25x80 mm. Larger granules produce more acetylene but increase reaction time. If granules of 8x15 and 15x25 mm decompose in 5-6 minutes, then granules of 25x80 mm require more than 10 minutes to decompose.
Historical reference
The first unusual carbon compound, similar to carbide, was obtained at the beginning of the 19th century by the Englishman Davy.
It was potassium carbide. Then, in 1863, unstable copper carbide was found, and 15 years later, iron carbide. Officially, the formations “appeared” only at the end of the 19th century - the Frenchman Henri Moisson had a hand in them. He made connections using a voltaic arc in an electric furnace, which he himself invented. For this purpose, charcoal heated to a red-hot state, pure metals and their oxides were used.
However, several years before Moisson, the mineral cohenite was discovered in meteorites - a mixture of cobalt, iron and nickel carbides. In a sense, this find helped answer the question “What are carbides?”
Welding applications
Carbide is stored in special steel tanks with a volume of 100 or 130 liters. These tanks should only be opened when there is no fire or sparks in the vicinity using a wooden hammer and a brass chisel. Unused carbide in the jar is sealed with a waterproof lid.
Acetylene for welding is produced from carbide in a generator of stationary or mobile type and of different volumes. The average volume of acetylene generators is designed to receive from 5 to 15 liters of water and, accordingly, 2-5 kilograms of carbide. The yield of acetylene is considered slightly lower than theoretical and is taken to be 260-280 liters per kilogram of CaC2. It is recommended to use coarse carbide – 80 mm
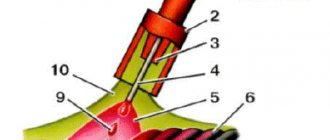
The principle of using acetylene for welding work is as follows:
- About 250 liters of acetylene are released from one kilogram of carbide, and 3-4 liters of water are required to decompose a kilogram of carbide. Knowing these proportions, the required volume of water and the amount of substance are calculated.
- In industrial generators, designed for a long and uniform process of use, carbide is dosed into the gas-forming chamber through a special hopper in automatic mode. In generators that are used for non-standardized volumes of work, the carbide is immersed in water in a special basket. The volume of acetylene produced is regulated by immersing or raising the basket.
- When the next portion of carbide is supplied and the reaction begins, the pressure in the chamber increases, which is reduced by the active release of acetylene into the burner.
- Acetylene is fed through a hose into the gas burner. The burner must be located at least 10 meters from the generator.
- The slaked lime formed during the reaction (about 1.2 kg per kilogram of carbide) is removed from the generator through a separate hopper.
In gas welding, the main advantage of using carbide is its low weight and the light weight of the equipment used. Gas cylinders for acetylene are very heavy; they must be moved on a special trolley or with 2-3 pairs of hands. The average generator weighs 15-20 kg, which allows you to move it without much effort alone or with one assistant. When moving dry carbide, it is enough to follow basic storage rules - avoid getting moisture on the substance and getting fine carbide dust on your skin and eyes.
Types of carbides
All substances can be divided into three groups:
- Covalent ones are formed by bromine and silicon; chemically inert, carbon atoms are in a state of sp-, sp2-, sp3-hybridization. It is formed by partial replacement of carbon atoms in diamond with bromine and silicon atoms. They have strong interatomic bonds, high melting points, heat resistance, chemical inertness, and are semiconductors. Bromine carbide is quite hard and can even scratch diamonds. Silicon carbide turns out to be more fragile, but chemically stable, oxidizes when exposed to oxygen only at temperatures above 1000 degrees Celsius, and dissolves in aqua regia and concentrated nitric acid and hydrofluoric acid.
- Salt-like or ionic are compounds formed by metals of groups I and II, as well as aluminum, rare earth elements and actinides. They are obtained directly from the elements or by reduction of carbon oxides. They are usually decomposed by water and acids, releasing a hydrocarbon and leaving a metal hydroxide. They have a high melting point.
- Metal-like or ionic-covalent-metallic are compounds that are formed by transition metals of groups IV–VII, as well as nickel, iron and cobalt; have intermediate chemical activity. In such compounds, small carbon atoms are located in the voids between the metal atoms. This is where the name interstitial carbides comes from. They are characterized by high strength and melting point.
In turn, ionic compounds are divided into:
- Methanides are usually transparent, colorless, and decompose in dilute acids and water to form methane. These include magnesium, aluminum and beryllium carbide.
- Acetylenides are actively hydrolyzed and form acetylene or ethylene. The best known is calcium carbide.
Safety precautions
Carbide for welding belongs to the class of explosive substances. Safe use of carbide is ensured by taking into account a number of conditions:
- Acetylene is a flammable gas, and dry carbide itself is also explosive, so there should be no open flames near the work site, even small ones such as lighters and lit cigarettes.
- It is prohibited to use carbide in granules up to 2 mm or carbide dust, as it dissolves very quickly, which leads to the release of large amounts of gas. Because of this, ultra-high pressure forms in the generator and an explosion may occur.
- It is prohibited to work with an angle grinder or an electric welding machine near gas welding operations and places where carbide drums are installed.
- The substance must be stored in a dry and sealed place, in which there are no water pipes, sewer pipes and, especially, gas equipment.
- Carbide dust upon contact causes irritation to the skin, eyes, and mucous membranes. Work with this substance must be carried out using personal protective equipment - goggles, gloves and a respirator.
- If carbide gets on your skin or eyes, rinse it with plenty of warm water and then carefully remove any remaining carbide with tweezers or a damp swab.
- When working with carbide in a workshop, all welding equipment must be placed in separate parts of the room, the ventilation system of the room must ensure the removal of flammable gases, and the room must be cleared of flammable materials.
- Acetylene generators are prohibited from being installed in basements and residential buildings.
- Before starting, the generator should be inspected for visible cracks or dents in the housing.
- During work, the generator must remain in a vertical position, the pressure gauge must be in working order and clearly visible to the welder or his assistant.
- At the end of the work, the carbide solution remaining in the generator must be completely exhausted, and the resulting lime must be disposed of.
- Reuse of wet carbide pieces is not permitted;
- Do not open the generator under pressure (during an ongoing reaction).
- Acetylene cylinders are stored and transported with special safety caps on the valves.
Compliance with these simple rules will ensure your own safety and the safety of people near the work site.
Carbide should be purchased from reputable welding equipment stores, since purchasing low-quality carbide can complicate welding work.
You can also order it in online stores. The price of carbide varies from 75 to 190 rubles per kilogram, depending on the manufacturer. If there is a need to regularly use equipment for gas welding and cutting, it is better to buy professional equipment manufactured at an industrial enterprise. The use of homemade generators can result in serious injuries and a threat to life.
Story
Metal carbides, the formulas of which we give below, are not natural compounds. This is due to the fact that their molecules tend to disintegrate when interacting with water. Therefore, it is worth talking here about the first attempts to synthesize carbides.
Since 1849, there have been references to the synthesis of silicon carbide, but some of these attempts remain unrecognized. Large-scale production began in 1893 by the American chemist Edward Acheson using a method that was later named after him.
The history of the synthesis of calcium carbide is also not very rich in information. In 1862, the German chemist Friedrich Wöhler obtained it by heating fused zinc and calcium with coal.
Now let's move on to more interesting sections: chemical and physical properties. After all, they contain the whole essence of the use of this class of substances.
Interesting connections
Sodium carbide. The formula of this compound is C2Na2. It can be thought of as an acetylenide (that is, the product of the replacement of hydrogen atoms in acetylene with sodium atoms) rather than a carbide. The chemical formula does not fully reflect these subtleties, so they must be looked for in the structure. This is a very active substance and upon any contact with water it very actively interacts with it to form acetylene and alkali.
Magnesium carbide. Formula: MgC2. Interesting ways to obtain this fairly active compound. One of them involves sintering magnesium fluoride with calcium carbide at high temperature. As a result, two products are obtained: calcium fluoride and the carbide we need. The formula for this reaction is quite simple, and you can read it in specialized literature if you wish.
If you are not sure of the usefulness of the material presented in the article, then the next section is for you.
Related materials[edit]
In addition to carbides, there are other groups of related carbon compounds: [2]
- graphite intercalation compounds
- alkali metal fullerides
- endohedral fullerenes, in which a metal atom is enclosed within a fullerene molecule
- metallacarbohedrenes (metcarbohedrenes) are cluster compounds containing C 2 units.
- tunable nanoporous carbon, in which gas chlorination of metal carbides removes metal molecules to form a highly porous, nearly pure carbon material capable of storing energy at high densities.
- carbene complexes of transition metals.
- two-dimensional transition metal carbides: MXenes
How can this be useful in life?
Well, firstly, knowledge of chemical compounds can never be superfluous. It is always better to be armed with knowledge than to be left without it. Secondly, the more you know about the existence of certain compounds, the better you understand the mechanism of their formation and the laws that allow them to exist.
Before moving on to the end, I would like to give some recommendations for studying this material.
Physical properties
When choosing almost any material, you should pay most attention to physical properties. For the one under consideration they are as follows:
- The compound has a crystalline structure.
- The melting point is 2300 °C. It is worth considering that such a figure is characteristic only of the pure composition. The addition of various impurities to the composition causes the melting point to drop significantly.
Pure Calcium Carbide
It is worth considering that calcium carbide in most cases is in a solid state. In addition, the color can vary from gray to brown. The physical properties of calcium carbide determine its widespread use in a wide variety of industries.