High-carbon steel, due to a number of undeniable advantages that it has, is successfully used for the production of products used in many industries. Meanwhile, the use of steels in this category is not always advisable, so it is very important to have a good understanding of the properties and quality characteristics of such alloys.
High carbon steel production
Compound
Depending on the amount of carbon, carbon and alloy steel are divided. The presence of carbon gives the material strength and hardness, and also reduces viscosity and ductility. Its content in the alloy is up to 2.14%, and the minimum amount of impurities due to the manufacturing process allows the bulk to consist of iron up to 99.5%.
High strength and hardness are what characterize carbon steel.
Impurities that are constantly included in the structure of carbon steel have a small content. Manganese and silicon do not exceed 1%, and sulfur and phosphorus are within 0.1%. An increase in the amount of impurities is characteristic of another type of steel, which is called alloyed.
The lack of technical ability to completely remove impurities from the finished alloy allows the following elements to be included in carbon steel:
- hydrogen;
- nitrogen;
- oxygen;
- silicon;
- manganese;
- phosphorus;
- sulfur
The presence of these substances is determined by the steel melting method: converter, open-hearth or other. And carbon is added on purpose. If the amount of impurities is difficult to regulate, then adjusting the level of carbon in the composition of the future alloy affects the properties of the finished product. When the material is filled with carbon up to 2.4%, steel is classified as carbon.
High carbon steel: characteristics, properties, grades and markings
High-carbon steel, due to a number of undeniable advantages that it has, is successfully used for the production of products used in many industries.
Meanwhile, the use of steels in this category is not always advisable, so it is very important to have a good understanding of the properties and quality characteristics of such alloys.
Features of the material
Any steel, as is known, is an alloy of iron and carbon, to which various alloying elements can be added.
The division of steels into low-, medium- and high-carbon types depends on the amount of carbon present in their composition.
This element, which has a serious impact on the characteristics of the finished alloy, can be contained in steels from 0.02 to 2.14%. In steels classified as high-carbon, the amount of this element in the composition starts at 0.6%.
Steel grades and the proportion of various elements in their composition
One of the distinctive features that high-carbon steels have is that products made from them are difficult to weld; welding leads to cracks appearing in the weld zone.
This is explained by the fact that such materials, having a certain chemical composition, have a tendency to form hardened zones in those places where the metal is exposed to thermal effects.
Due to this feature of high-carbon steels, welding products made from them should only be done using electrodes with low thermal power. The welding arc used to connect products made of high-carbon steels must be of the recovery type.
The use of an oxidizing arc in such cases will lead to the fact that carbon will be burned out of the steel composition, and, as a result, the metal in the weld area will become more porous. Meanwhile, such a negative effect can be avoided if the products to be joined are preheated to a temperature of 200–2500.
Characteristic
The characteristics and structure of the metal are changed using heat treatment, through which the required surface hardness or other requirements for the use of the steel structure are achieved. However, not all structural properties can be adjusted using thermal methods. Such structurally insensitive characteristics include rigidity, expressed by the elastic modulus or shear modulus. This is taken into account when designing critical components and mechanisms in various fields of mechanical engineering.
In cases where the calculation of the strength of an assembly requires the use of small-sized parts that can withstand the required load, heat treatment is used. This effect on “raw” steel makes it possible to increase the rigidity of the material by 2-3 times. The metal that is subjected to this process is subject to requirements regarding the amount of carbon and other impurities. This steel is called high quality.
Formation of cementite when heating high-carbon steel
For example, consider high-carbon steel with a carbon content of 0.95%. If this steel is heated to 760 ºС, then its point on the phase diagram of Figure 1 will be a white circle with horizontal arrows passing through it. Since this point lies in the two-phase region γ + Acm, the steel should consist of a mixture of austenite of composition O (0.85% carbon) and cementite (0.67% carbon). The diagram does not give a view of the microstructure, but experiments show that it appears as shown at the bottom of Figure 1. All cementite is in the form of small spherical grains, which are randomly distributed among the austenite grains. The sizes of austenite grains are much larger than cementite grains. Austenite grains have clear boundaries.
Classification of carbon steels
According to the direction of application of products, carbon steel is divided into tool and structural.
The last of them is used for the construction of various buildings and frame parts. Tools are used to make durable tools for performing any work, including metal cutting. The use of metal products in the household required the classification of steel into different categories with specific properties: heat-resistant, cryogenic and corrosion-resistant.
According to the method of production, carbon steels are divided into:
- electric steel;
- open hearth;
- oxygen converter.
Differences in the structure of the alloy are due to the presence of different impurities characteristic of a particular smelting method.
The relationship of steel to chemically active environments has made it possible to divide products into:
- boiling;
- semi-calm;
- calm.
Carbon content divides steel into 3 categories:
- hypereutectoid, in which the amount of carbon exceeds 0.8%;
- eutectoid, with a content of 0.8%;
- hypoeutectoid – less than 0.8%.
It is the structure that is a characteristic feature in determining the state of the metal. In hypoeutectoid steels, the structure consists of pearlite and ferrite. Eutectoid ones have pure pearlite, while hypereutectoid ones are characterized by pearlite with admixtures of secondary cementite.
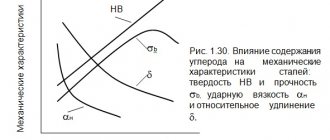
By increasing the amount of carbon, steel increases strength and reduces ductility. The viscosity and brittleness of the material also have a great influence. As the percentage of carbon increases, the impact strength decreases and the fragility of the material increases. It is no coincidence that when the content is more than 2.4%, metal alloys are already classified as cast iron.
According to the amount of carbon in the alloy, steel is:
- low carbon (up to 0.29%);
- medium carbon (from 0.3 to 0.6%);
- high carbon (more than 0.6%).
High-quality carbon steel for structural purposes
They are the main metal for the manufacture of machine parts (shafts, spindles, axles, gears, keys, couplings, flanges, friction discs, screws, nuts, stops, rods, hydraulic cylinders, eccentrics, chain sprockets, etc.), which, when interacting in a working machine, they perceive and transmit loads of varying magnitude. These metals are well processed by pressure and cutting, cast and welded, and are subjected to thermal, thermomechanical and chemical-thermal treatment.
Various special types of processing provide the toughness, elasticity and hardness of steels, making it possible to make parts from them that are viscous in the core and hard on the outside, which dramatically increases their wear resistance and reliability. High-quality carbon structural steels are used to produce rolled products, forgings, calibrated steel, silver steel, long steel, stampings and ingots.
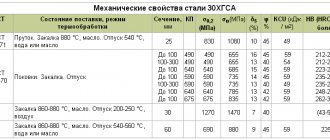
Table 3. Basic properties of high-quality carbon structural steel
| Brand | Mechanical properties | Physical properties | Technological properties | ||||||||||
| σт | σв | δ, % | an J/cm2 | NV | γ, g/cm3 | λ, W/(m °С) | α·106.1/°С | treatments- productivity cutting | weldability _ | interval temperatures forging, °C | plastic at cold processing | *hot- rolled **identify- married | |
| MPa | |||||||||||||
| 08 | 196 | 324 | 33 | — | 126 | 7,83 | 811 | 11,6 | IN | BB | 800-1300 | BB | * |
| 10 | 206 | 321 | 31 | — | 140 | 7,83 | 811 | 11,6 | IN | BB | 800-1300 | BB | * |
| 15 | 225 | 373 | 27 | — | 145 | 7,82 | 770 | 11,9 | IN | BB | 800-1250 | BB | * |
| 20 | 245 | 412 | 25 | — | 159 | 7,82 | 770 | 11,1 | IN | BB | 800-1280 | IN | * |
| 25 | 274 | 451 | 23 | 88 | 166 | 7,82 | 732 | 11,1 | IN | BB | 800-1280 | IN | * |
| 30 | 294 | 490 | 21 | 78 | 175 | 7,817 | 732 | 12,6 | IN | IN | 800-1250 | IN | * |
| 35 | 314 | 529 | 20 | 69 | 203 | 7,817 | 732 | 11,09 | IN | IN | 800-1250 | IN | * |
| 40 | 321 | 568 | 19 | 59 | 183 | 7,815 | 596 | 12,4 | IN | U | 800-1250 | U | ** |
| 45 | 363 | 598 | 16 | 49 | 193 | 7,814 | 680 | 11,649 | IN | U | 800-1250 | U | ** |
| 50 | 373 | 627 | 14 | 38 | 203 | 7,811 | 680 | 12,0 | U | U | 800-1250 | U | ** |
| 55 | 382 | 647 | 13 | — | 212 | 7,82 | 680 | 11,0 | U | N | 800-1250 | N | ** |
| 60 | 402 | 676 | 12 | — | 224 | 7,80 | 680 | 11,1 | U | N | 800-1240 | N | ** |
| Note. N - low, U - satisfactory, V - high, BB - very high. | |||||||||||||
High-quality structural steels have higher mechanical properties (GOST 1050-88) than steels of ordinary quality, due to their lower content of phosphorus, sulfur and non-metallic inclusions. According to the type of processing, they are divided into hot-rolled, forged, calibrated and silver (with special surface finishing).
The designation of the steel grade is made up of the word “Steel” and a two-digit number that indicates the average carbon content in hundredths of a percent. For example, Steel 25 contains 0.25% carbon (permissible amount of carbon - 0.220.30%), Steel 60-0.60% (permissible amount -0.57-0.65%). The degree of deoxidation is not reflected in grades of calm steels, but in grades of semi-quiet and boiling steels, as well as steels of ordinary quality, it is designated by the letters “ps” and “kp”, respectively. In high-quality structural steels of all grades, the sulfur content is allowed to be no more than 0.040% and phosphorus - no more than 0.035%.
The main properties of high-quality carbon structural steel are given in table. 3, main purpose - in table. 4. Marking colors are given in table. 5.
Table 4. High-quality carbon steel for structural purposes, their main purpose
| steel grade | Main purpose |
| Steel 08kp, 10 | Parts produced by cold stamping and cold heading, tubes, gaskets, fasteners, caps. Cemented and cyanidated parts that do not require high core strength (bushings, rollers, stops, copiers, gears, friction discs) |
| Steel 15, 20 | Lightly loaded parts (rollers, pins, stops, copiers, axles, gears). Thin parts exposed to abrasion, levers, hooks, traverses, liners, bolts, couplers, etc. |
| Steel 30, 35 | Parts experiencing low stress (axles, spindles, sprockets, rods, traverses, levers, disks, shafts) |
| Steel 40, 45 | Parts that require increased strength (crankshafts, connecting rods, ring gears, camshafts, flywheels, gears, pins, ratchets, plungers, spindles, friction discs, axles, couplings, racks, rolling rollers, etc.) |
| Steel 50, 55 | Gear wheels, rolling rollers, rods, bandages, shafts, eccentrics, lightly loaded springs and leaf springs, etc. Used after hardening with high tempering and in a normalized state |
| Steel 60 | Parts with high strength and elastic properties (rolling rolls, eccentrics, spindles, spring rings, clutch springs and discs, shock absorber springs). Apply after hardening or after normalization (large parts) |
Table 5. Marking colors for high-quality carbon steel
| Group | Paint color |
| Steel 08, 10, 15, 20 | White |
| Steel 25, 30, 35, 40 | White and yellow |
| Steel 45, 50, 55, 60 | White and brown |
Marking
When designating carbon steels of ordinary quality, the letters St are used, which are accompanied by numbers characterizing the carbon content. One digit shows the quantity multiplied by 10, and two digits by 100. When guaranteeing the mechanical composition of the alloy, B is added before the designation, and compliance with the chemical constituents is B.
At the end of the marking, two letters indicate the degree of deoxidation: ps - semi-quiet, kp - boiling state of the alloys. For calm metals this indicator is not indicated. An increased amount of manganese in the structure of the product is designated by the letter G.
When designating high-quality carbon steels used in the manufacture of tools, the letter U is used, next to which a number is written confirming the percentage of carbon in a 10-fold amount, regardless of whether it is two-digit or single-digit. To highlight higher quality alloys, the letter A is added to the designation of tool steels.
Examples of designation of carbon steels: U8, U12A, St4kp, VSt3, St2G, BSt5ps.
Phase diagram for hypereutectoid steels
Figure 1 shows the iron-carbon phase diagram, including the part with a high carbon content - for hypereutectoid - high-carbon - steels. Cementite plays an important role here.
Figure 1 – Iron-carbon phase diagram, including a section of hypereutectoid (high-carbon) steels (carbon content more than 0.77%)
The region where austenite exists is shown as a dark central region. Because cementite exists in only one chemical composition, it is shown as a vertical line in the diagram at 6.7% carbon.
Production
The metallurgical industry produces metal alloys. The specificity of the process for producing carbon steel is the processing of cast iron billets with the reduction of suspended matter such as sulfur and phosphorus, as well as carbon, to the required concentration. The differences in the oxidation technique by which carbon is removed allows us to distinguish different types of smelting.
Oxygen converter method
The basis of the technique was the Bessemer method, which involves blowing air through liquid cast iron. During this process, carbon is oxidized and removed from the alloy, after which the iron ingots gradually turn into steel. The productivity of this technique is high, but sulfur and phosphorus remained in the metal. In addition, carbon steel is saturated with gases, including nitrogen. This improves strength but reduces ductility, making the steel more prone to aging and high in non-metallic elements.
Given the low quality of steel produced by the Bessemer method, it was no longer used. It was replaced by the oxygen-converter method, the difference of which is the use of pure oxygen, instead of air, when purging liquid cast iron. The use of certain technical conditions during purging significantly reduced the amount of nitrogen and other harmful impurities. As a result, carbon steel produced by the oxygen-converter method is close in quality to alloys melted in open-hearth furnaces.
The technical and economic indicators of the converter method confirm the feasibility of such smelting and make it possible to replace outdated methods of steel production.
Open hearth method
A feature of the method for producing carbon steel is the burning of carbon from cast iron alloys not only with the help of air, but also by adding iron ores and rusty metal products. This process usually takes place inside furnaces, to which heated air and combustible gas are supplied.
The size of such melting baths is very large; they can hold up to 500 tons of molten metal. The temperature in such containers is maintained at 1700 ºC, and carbon burning occurs in several stages. First, due to excess oxygen in flammable gases, and when slag forms above the molten metal, through iron oxides. When they interact, slags of phosphates and silicates are formed, which are subsequently removed and the steel acquires the required quality properties.
Steel melting in open hearth furnaces takes about 7 hours. This allows you to adjust the desired composition of the alloy when adding different ores or scrap. Carbon steel has long been manufactured using this method. Such stoves, in our time, can be found in the countries of the former Soviet Union, as well as in India.
Electrothermal method
It is possible to produce high-quality steel with a minimum content of harmful impurities by melting it in vacuum furnaces of electric arc or induction furnaces. Thanks to the improved properties of electric steel, it is possible to produce heat-resistant and tool alloys. The process of converting raw materials into carbon steel occurs in a vacuum, due to which the quality of the resulting workpieces will be higher than the previously discussed methods.
The cost of such metal processing is more expensive, so this method is used when there is a technological need for a high-quality product. To reduce the cost of the technological process, a special ladle is used, which is heated inside a vacuum container.
Production methods and quality division options
The production of carbon steels is based on the processing of cast iron and ferrous scrap with the reduction of sulfur, phosphorus and carbon to the required concentration. At the same time, various technologies are used, which affect the quality of the metal produced. As a result, the following types of steel alloys are obtained:
- high quality;
- quality;
- ordinary quality.
The first two types of carbon steels are produced using converters, open-hearth furnaces and more modern electric melting furnaces. The chemical composition of such steels and the concentration of impurities contained are strictly regulated by GOSTs 1435–99 and 1050–88. Thus, for high-quality steel alloys, sulfur can be used in an amount of no more than 0.04%, phosphorus - no more than 0.035%, for high-quality steel alloys, respectively - less than 0.018% and no more than 0.025%. Carbon steels of these categories have a structure of increased purity.
The smelting of steel alloys of ordinary quality is carried out on the basis of oxygen converters and open-hearth furnaces, then large ingots are formed from them. In comparison with the previous two categories, carbon steels of ordinary quality are characterized by the content of a large amount of harmful impurities. According to GOST 380–2005, they contain up to 0.05% sulfur and up to 0.04% phosphorus. Such material is subjected to hot and cold rolling in order to produce thin- and thick-sheet material and wide metal strips.
The purpose of heat treatment of steel is to change its microstructure
It is impossible not to see a significant difference between the microstructure in Figure 2 and that shown in Figure 1 - after heating the steel from room temperature to 760 ºC. Note that both microstructures contain the same amount of cementite and austenite, but the distribution of cementite is completely different.
Unlike austenite and ferrite, cementite is very brittle. Therefore, steel with the microstructure in Figure 2, with interconnected cementite plates, will not be as tough and ductile as steel with the microstructure in Figure 1 with small, isolated cementite grains. These cementite grains give the steel good cutting properties. For example, straight razors are made from such steels.
This is a good example of how heat treating steel - not just high-carbon steel - can change its microstructure and therefore its mechanical properties.
Note that the microstructures that are shown in Figures 1 and 2 only exist at a high temperature of 760 ºC, and not at room temperature. Phases and microstructures at room temperature are in a separate article.